Home
The central goals of the Resource for Integrated Glycotechnology are to increase understanding of the molecular and functional roles of protein-carbohydrate interactions and develops more powerful technologies necessary to achieve these goals.
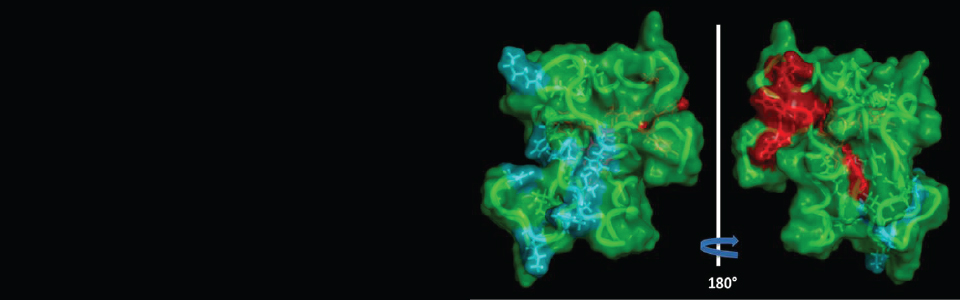
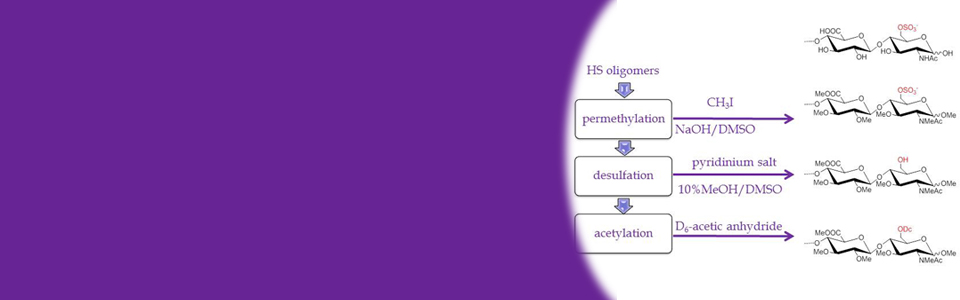
Complex carbohydrates play an important role in many biomedically important processes, including inflammatory response, hormone action, malignancy, viral and bacterial infections and cell differentiation. This Resource is focused on the developing a novel, integrated toolkit of technologies that will address the challenges of determining the structures of carbohydrates, details of their interactions with binding proteins, and elucidation of their biological functions by leveraging advances in analytical, synthetic, structural, biochemical and biological tools. Three broad Technology Research and Development (TR&D) projects are being pursued in concert with a collection of Driving Biomedical Projects (DBPs). These latter projects act as test bed platforms for assessing the utility of the integrated approaches and provide feedback for new challenges that will spur further technology development.
As the technologies mature they are being applied to a collection of Collaborative Projects and used in Analytical Services that extend the utility of the technologies to the broader scientific community. Multiple strategies for Dissemination, including this website, will increase awareness and access to technology developments. Training courses provide direct opportunities for researchers to learn the latest technology developments from Resource staff.
The Resource for Integrated Glycotechnology is supported by a grant from the National Institute of General Medical Sciences (GM103390) of the National Institutes of Health.